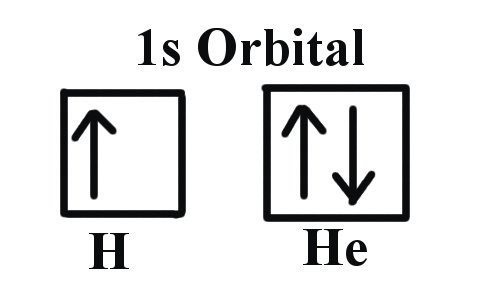
Pauli’s Principle of Exclusion in the Field of Chemistry He contributed his great time and efforts to the field of quantum mechanics and took it to a new height. In 1945, he was awarded the Nobel prize for his discovery of the principle named after his last name, Pauli’s principle of exclusion. They were already based on a constructed definition under his own spin-statistic theorem, and he described fermions using his principle, which included neutrinos and electrons. A few years later, in 1940, he expanded his research and came up with the facts about bosons and fermions stated above. He basically studied electron behaviour and came up with this principle to describe the same. This principle and this term were coined in the year 1925 by an Australian physicist named, Wolfgang Pauli. This theory eventually considered all the possible and ready-to-be conceived information about electrons and shell structures and did matter with the quantum shell sets. Regarding the expected results from the experiment, he concluded that a complicated number of electrons in any shell can be simplified and can be brought down to a simple, smaller number that will be easy to understand. After this, Pauli expanded his area of research and looked for a solution that was factual. Later, a theory stated that after a certain number of electrons in any shell, that shell is eventually considered a closed shell. These many theories basically state the statements as assumptions regarding the number of maximum electrons occupying a specific number of electron shells. There were many theories that came into the bigger picture in the late years of the 1900s. It was a clear fact, even in the 1900s, that molecules and atoms are more stable when they have an even number of electrons than when they have an odd number of electrons. Where it goes with some particles with a half-spin feature, it somehow doesn’t go with all the particles that have an integral spin, such as bosons. These fermions are named after the data-driven nature they possess. This principle doesn’t only restrict itself to electrons, but it is also considered with other particles too, like fermions that have half-integral spin. When we say that it cannot have the same set for all four quantum numbers, we mean n, l, ml, and ms. In other words, it states that no two or more than two electrons can have the same four quantum numbers in its set and they can’t be identical either. Pauli’s principle of exclusion is a straightforward fact that states that any orbit can have a maximum of two electrons, and those electrons should be of the opposite spin.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
